Photo: ChatGPT
Introduction
Coral reefs, which are crucial to the health and diversity of the global marine ecosystem, have experienced a considerable decline over the past 2 decades due to human activities and climate change. According to the Status of Coral Reefs of the World: 2020 report, approximately 14% of global coral reefs were lost between 2009 and 2018 due to rising ocean temperatures, acidification, and pollutionv 1[1]. Frequent coral bleaching events, driven by prolonged increasing sea temperatures, have had devastating effects on reef ecosystems 2[2]. Mass coral bleaching episodes in 1998, 2010, and 2015 collectively impacted over 70% of coral reefs worldwide, underscoring the urgency of conservation strategies 3[3,4]. Be 56Cryopreservation, a method for preserving biological materials at low temperatures to ensure their vital functions and viability remain intact upon thawing5,6,7], has been employed on a variety of substances, such as coral gametes and fish genetic material 8[8,9]. This technique holds promise for the conservation of endangered species and for facilitating advancements in developmental biology and marine biotechnology [10,11]. Despite its potential, cryopreservation poses formidable challenges. For example, ice crystals 13may form [12,13], which may result in damage to cells and tissues and increase the risk of contamination by pathogenic agents [14].Vitrification, an advanced cryopreservation method, rapidly cools biological samples to freezing temperatures in a glass-like medium, preventing ice crystal formation. Although vitrification is not yet widely established in aquatic cryopreservation, it has been experimentally applied to a limited number of invertebrate species and offers potential advantages over slow-cooling methods, including simplified equipment requirements and shorter processing times 8[8].
Materials and methodsv
Coral collection and oocyte isolation
The specimens J. fragilis and J. juncea were collected during their reproductive season (June–September) from Kenting National Park, Nanwan, Taiwan (21°56’N, 120°44’E) at depths of 3–10 m. The National Kenting Park Management Office of Taiwan issued a coral collection permit to the study authors. Professional divers subsequently collected the permitted quantities of J. fragilis and J. juncea branches, which were then transported to the coral husbandry center of the National Museum of Marine Biology and Aquarium in Checheng, Taiwan. Upon arrival, the branches J. fragilis and J. junceawere affixed to substrates with bindings and placed in half-ton tanks with gravel bases. Native seawater with a salinity of 35 ppt was circulated through the tanks, which were maintained at a constant temperature of 25°C. Each branch measured approximately 60 cm and was analyzed and categorized by oocyte type and quantity before being transferred to the husbandry facility.
Effects of vitrification solution on oocytes
The concentrations of CPAs used in the production of Equilibration Solution 1 (ES1), ES2, and VS are presented in Table 1. These solutions sequentially prepared the oocytes for exposure to the VS. This experimental phase was used to evaluate the toxicity of ES1 (1 M propylene glycol (PG) + 0.25 M ethylene glycol (EG) + 0.5 M methanol), ES2 (2 M PG + 0.5M EG + 1 M methanol), and VS (4 M PG + 1 M EG + 2 M methanol) to the oocytes. In each experiment, 30–50 freshly harvested oocytes were immersed in one of the three solutions in a six-well culture dish. For ES1 and ES2, five oocytes were removed at three intervals: 5, 10, and 15 minutes. ATP counts were used to determine oocyte viability. Five oocytes were subjected
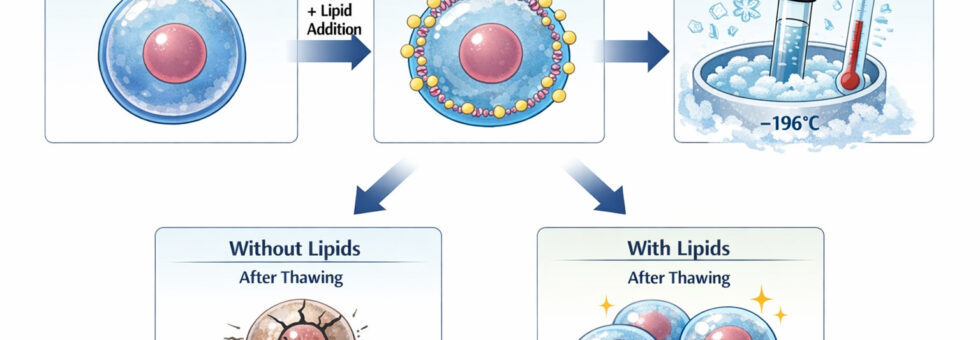
Incorporation of liposomes into VS for vitrification
o investigate the effect of lipid composition on oocyte cryotolerance, nine lipid types were selected based on their structural roles in cellular membranes and previously reported cryoprotective properties in gametes and embryos. The lipids included phospholipids (PLs: phosphatidylcholine (PC) and phosphatidylethanolamine (PE), monounsaturated fatty acids (MUFAs: erucic acid, oleic acid, palmitoleic acid), and polyunsaturated fatty acids (PUFAs: docosahexaenoic acid, linoleic acid, and eicosatrienoic acid).PLs such as PC and PE are integral to coral oocyte membranes and influence membrane integrity under low-temperature conditions. MUFAs were chosen for their ability to maintain membrane fluidity and reduce chilling injury, while PUFAs were included due to their reported benefits in vertebrate cryopreservation models, although their effects in marine invertebrates remain uncertain.
Source:
Press release link: Incorporation of lipids improves cryo-tolerance of vitrified gorgonian coral oocytes
Autores: Yen-Po Chen, Tzu-Fei Huang, Ester Lo, Sujune Tsai, Zhi-Hong Wen, Hung-Kai Chen, Chiahsin Lin
Photo ©: ChatGPT
Copyright © 2026 Smithsonian. All rights reserved.